Chair conformation benzene Many compounds discovered in the nineteenth century seemed to be related to benzene. Structure and conformations of decalins.
Chair Conformation Benzene, The distance from atom 1 to atom 4 depends on the absolute value of the dihedral angle. Cyclic alkanes can also interconvert between their conformers by rotation of the single carbon to carbon bonds. Benzene has six hydrogen atoms.
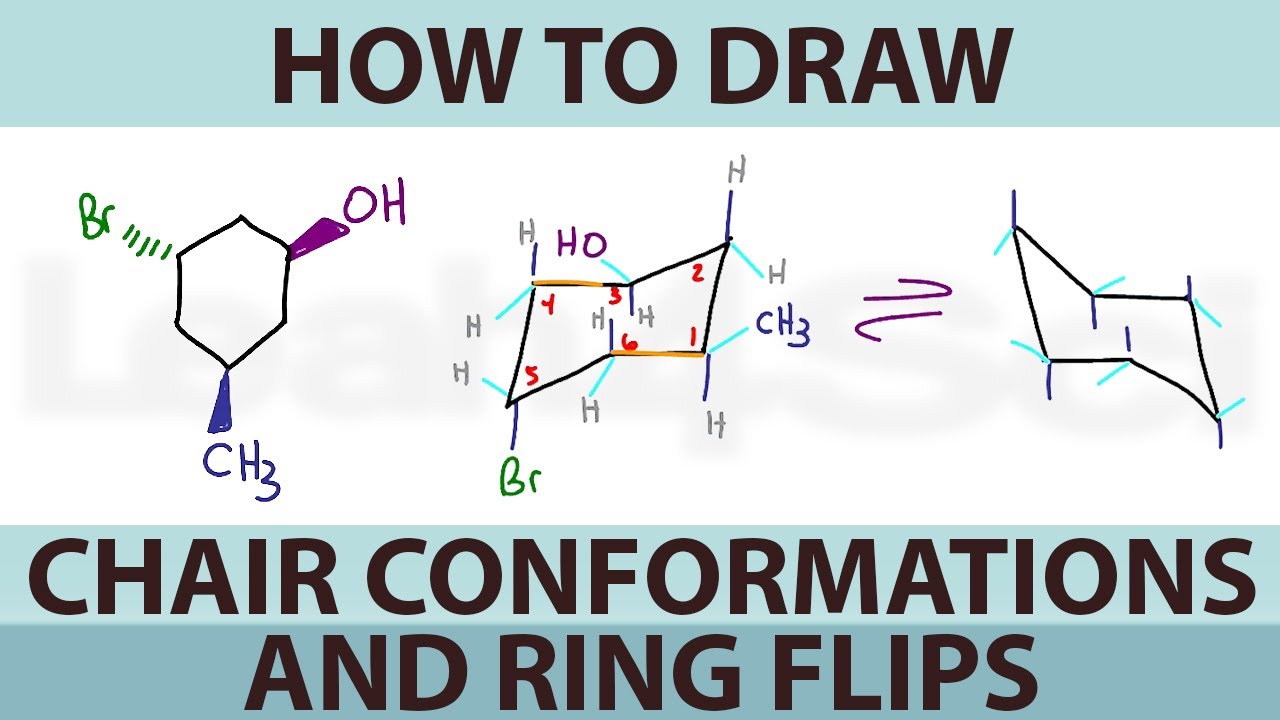 How To Draw Cyclohexane Chair Conformations And Ring Flips Youtube From youtube.com
How To Draw Cyclohexane Chair Conformations And Ring Flips Youtube From youtube.com
Another Article :
Moreover the symmetry of chair conformation is D 3d while boat symmetry has the symmetry C 2v. The distance from atom 1 to atom 4 depends on the absolute value of the dihedral angle. If yours looks a little sloppy consider grabbing The Pocket Chemist molecule stencil below to draw quickly draw your chair and boat conformations benzene rings bonding angles perfectly every time. It is important to remember that the bonds on a given side of a chair ring-conformation always alternate in this fashion. This is your first axial substituent.
The molar mass of cyclohexane is 8416 g mol.
Because of the alternating nature of equatorial and. Get the drawing done in a third of the time so you can focus on the difficult parts of the reactions. Therefore it should be clear that for cis-12-disubstitution one of the substituents must be equatorial and the other axial. Other organic compounds without these properties were called aliphatic meaning fatlike. Moreover the symmetry of chair conformation is D 3d while boat symmetry has the symmetry C 2v.
 Source: differencebetween.com
Source: differencebetween.com
Difference Between Chair And Boat Conformation Compare The Difference Between Similar Terms Chair Conformation In the chair conformation there are two planes with carbon atoms alternating making carbon atoms 1 3 and 5 in one plane and carbon atoms 2 4 and 6 in another plane. Ive seen chair conformation defined in that way before. These compounds also had low hydrogen-to-carbon ratios as well as pleasant aromas. Boat twist boat and half-chair conformations. Get the drawing done in a third of the time so you can focus on the difficult parts of the reactions. The chair conformation of cyclohexane is not rigid.
 Source: youtube.com
Source: youtube.com
How To Draw Cyclohexane Chair Conformations And Ring Flips Youtube Besides boat conformation tends to convert into the boat-twist conformation. Chair Conformation In the chair conformation there are two planes with carbon atoms alternating making carbon atoms 1 3 and 5 in one plane and carbon atoms 2 4 and 6 in another plane. These two chair conformations are the most common and comfortable of all the conformational possibilities available to a cyclohexane ring. The molar mass of cyclohexane is 8416 g mol. They both have different molecular mass melting point and boiling point. Of cyclohexane occupy coplanar positions and when carbon atoms 3 and 6 are on opposite sides of the aircraft conformation from the D3d symmetry group is called a form of chair.
![]() Source: biorender.com
Source: biorender.com
Biorender Life Science Icons Many compounds discovered in the nineteenth century seemed to be related to benzene. The chair and twist-boat are energy minima and are therefore conformers while the half-chair and the boat are transition states and represent energy maxima. The molar mass of cyclohexane is 8416 g mol. These two chair conformations are the most common and comfortable of all the conformational possibilities available to a cyclohexane ring. Cyclohexane has chair conformation. The chair conformation has alternating axial up axial down so once you have that single axial substituent move on to.
 Source: sciencedirect.com
Source: sciencedirect.com
Ring Flipping An Overview Sciencedirect Topics Different spatial arrangements of atoms that result from rotations about single σ bonds. These two chair conformations are the most common and comfortable of all the conformational possibilities available to a cyclohexane ring. Identify the up tip OR down tip of your chair conformation and draw a straight line up up tip or down down tip parallel to the y-plane. The energy barriers between the chair boat and twist conformations of cyclohexane are low enough Fig6 to make separation of the. Therefore it should be clear that for cis-12-disubstitution one of the substituents must be equatorial and the other axial. Ive seen chair conformation defined in that way before.
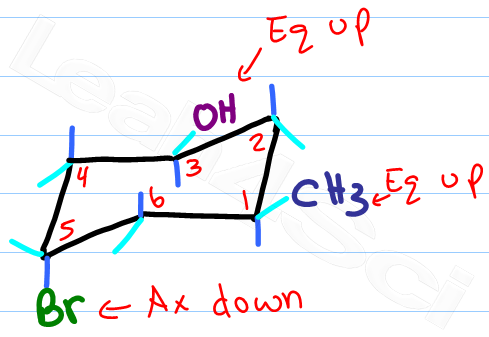 Source: leah4sci.com
Source: leah4sci.com
Drawing Chair Conformations And Ring Flips For Cyclohexane In Organic Chemistry Benzene has sp2 hybridized carbon atoms and cyclohexane has sp3 hybridised carbon atoms. This is true for 1R-33-dichlorocyclohexanol. That is the specific structure for cyclohexane. Cyclohexane has chair conformation. They both have different molecular mass melting point and boiling point. So the 13-diaxial notation is the most common way we refer to the gauche interactions of axial groups in the chair conformations.
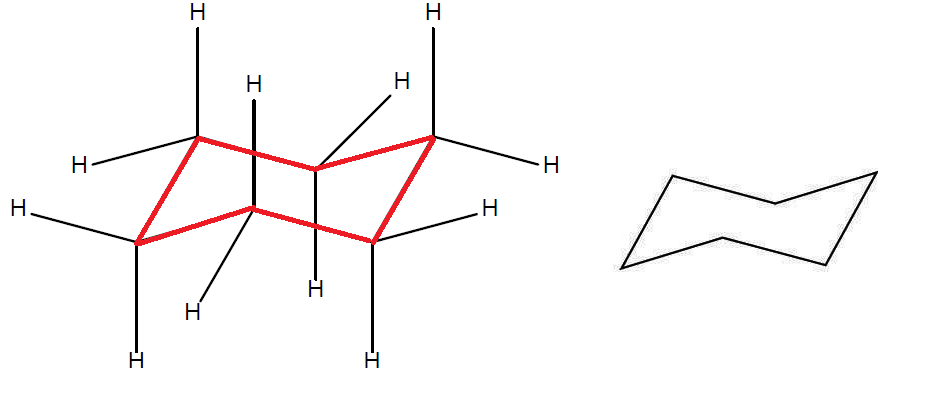 Source: documentation.samson-connect.net
Source: documentation.samson-connect.net
Chair Boat And Twist Boat Conformation Of Cyclohexanes And Glucose Generally the axial conformation of a given cyclohexane is less stable than the corresponding equatorial conformation. Boat twist boat and half-chair conformations. Apart from the number of hydrogen atoms benzene has a planar structure and cyclohexane has a chair confirmation. We can think of it as two chains mirror images one of the other containing atoms 1-2-3-4 and 1-6-5-4 with opposite dihedral angles. Click the structures and reaction arrows to view the 3D models and animations respectively. 95 13-diaxial interactions Methylcyclohexane equatorial Methylcyclohexane axial gauche.
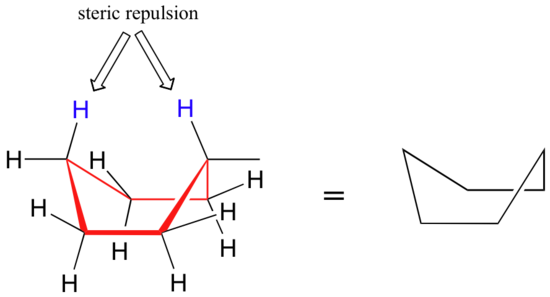 Source: chem.libretexts.org
Source: chem.libretexts.org
3 2 Conformations Of Cyclic Organic Molecules Chemistry Libretexts In the trans-isomer both may be equatorial. This is your first axial substituent. Usually chair conformation is the most stable conformation and at room temperature about 9999 of cyclohexane in a mixture of different conformation exists in this conformation. So the 13-diaxial notation is the most common way we refer to the gauche interactions of axial groups in the chair conformations. This group of compounds was called aromatic because of their pleasant odors. It can convert to a twist boat comformation and then to a new chair conformation in a process termed ring-flipping as shown Figure 614 not all the hydrogens are shown for clarity.
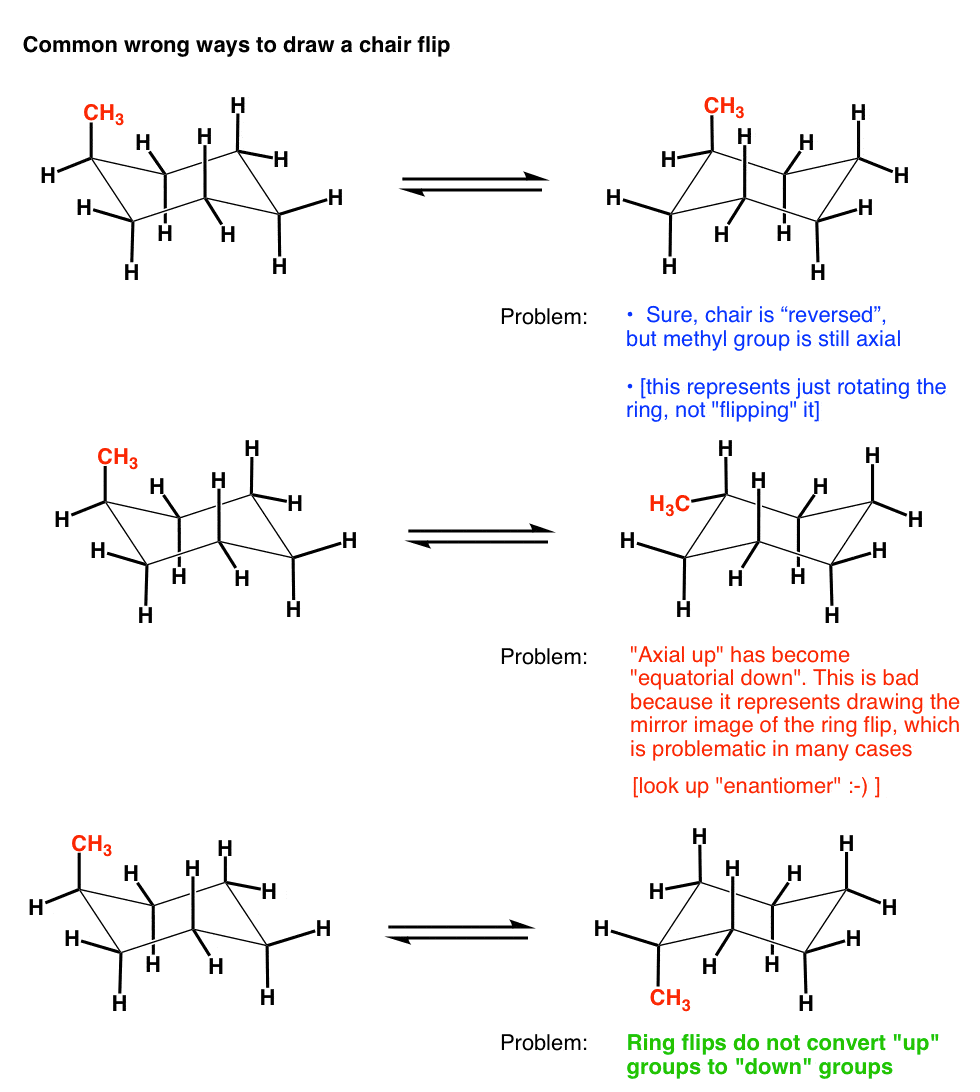 Source: masterorganicchemistry.com
Source: masterorganicchemistry.com
The Cyclohexane Chair Flip Master Organic Chemistry Benzene is a planar structure. In the ring-flipping process C. Identify the up tip OR down tip of your chair conformation and draw a straight line up up tip or down down tip parallel to the y-plane. In the trans-isomer both may be equatorial. Almost all of your work with cyclohexanes will involve chair conformations. Because of the alternating nature of equatorial and.
 Source: leah4sci.com
Source: leah4sci.com
Drawing Chair Conformations And Ring Flips For Cyclohexane In Organic Chemistry Ive seen chair conformation defined in that way before. The idea that the chair conformation is the most stable structure for cyclohexane was first proposed as early as 1890 by Hermann Sachse but only gained widespread acceptance much later. It can convert to a twist boat comformation and then to a new chair conformation in a process termed ring-flipping as shown Figure 614 not all the hydrogens are shown for clarity. Because of the alternating nature of equatorial and. Therefore it should be clear that for cis-12-disubstitution one of the substituents must be equatorial and the other axial. The same term applies to similar conformations of six-member saturated ring analogous structures.
 Source: researchgate.net
Source: researchgate.net
Chemical Structures Of Benzene Top And Cyclohexane Bottom Benzene Download Scientific Diagram In the trans-isomer both may be equatorial. It can convert to a twist boat comformation and then to a new chair conformation in a process termed ring-flipping as shown Figure 614 not all the hydrogens are shown for clarity. Identify the up tip OR down tip of your chair conformation and draw a straight line up up tip or down down tip parallel to the y-plane. They both have different molecular mass melting point and boiling point. The energy barriers between the chair boat and twist conformations of cyclohexane are low enough Fig6 to make separation of the. This group of compounds was called aromatic because of their pleasant odors.
 Source: researchgate.net
Source: researchgate.net
Configuration And Preferred Half Chair Conformation Of The Two Download Scientific Diagram The molar mass of cyclohexane is 8416 g mol. We can think of it as two chains mirror images one of the other containing atoms 1-2-3-4 and 1-6-5-4 with opposite dihedral angles. This group of compounds was called aromatic because of their pleasant odors. Besides boat conformation tends to convert into the boat-twist conformation. The molar mass of cyclohexane is 8416 g mol. Click the structures and reaction arrows to view the 3D models and animations respectively.
 Source: shefalitayal.com
Source: shefalitayal.com
Cyclohexane Conformer The Cyclohexane Chair Flip Part B Cyclohexane Exists In Different Conformers Cyclohexane Stereochemistry We can think of it as two chains mirror images one of the other containing atoms 1-2-3-4 and 1-6-5-4 with opposite dihedral angles. The molar mass of cyclohexane is 8416 g mol. The molar mass of benzene is. The idea that the chair conformation is the most stable structure for cyclohexane was first proposed as early as 1890 by Hermann Sachse but only gained widespread acceptance much later. In a chair conformation with a few exceptions only axial substituents will give rise to steric strain energy. Boat and Twist Boat Conformations There are additional conformations of cyclohexane rings.
 Source: pediaa.com
Source: pediaa.com
Difference Between Cyclohexane And Benzene Definition Chemical Properties Structure Synthesis And Differences Boat twist boat and half-chair conformations. Therefore it should be clear that for cis-12-disubstitution one of the substituents must be equatorial and the other axial. In the trans-isomer both may be equatorial. Get the drawing done in a third of the time so you can focus on the difficult parts of the reactions. The molar mass of cyclohexane is 8416 g mol. The same term applies to similar conformations of six-member saturated ring analogous structures.
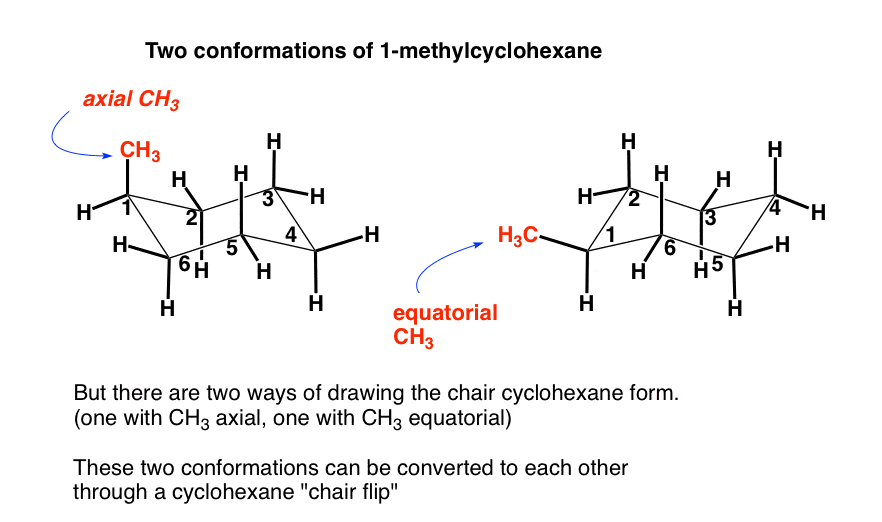 Source: masterorganicchemistry.com
Source: masterorganicchemistry.com
The Cyclohexane Chair Flip Master Organic Chemistry The same term applies to similar conformations of six-member saturated ring analogous structures. If yours looks a little sloppy consider grabbing The Pocket Chemist molecule stencil below to draw quickly draw your chair and boat conformations benzene rings bonding angles perfectly every time. In the trans-isomer both may be equatorial. This group of compounds was called aromatic because of their pleasant odors. We can think of it as two chains mirror images one of the other containing atoms 1-2-3-4 and 1-6-5-4 with opposite dihedral angles. Benzene has sp2 hybridized carbon atoms and cyclohexane has sp3 hybridised carbon atoms.
![]() Source: biorender.com
Source: biorender.com
Biorender Life Science Icons Cyclic alkanes can also interconvert between their conformers by rotation of the single carbon to carbon bonds. Ive seen chair conformation defined in that way before. Generally the axial conformation of a given cyclohexane is less stable than the corresponding equatorial conformation. We can think of it as two chains mirror images one of the other containing atoms 1-2-3-4 and 1-6-5-4 with opposite dihedral angles. The chair conformation is estimated to be lower in energy than the twist conformation by approximately 23 kJ mol-1. The idea that the chair conformation is the most stable structure for cyclohexane was first proposed as early as 1890 by Hermann Sachse but only gained widespread acceptance much later.
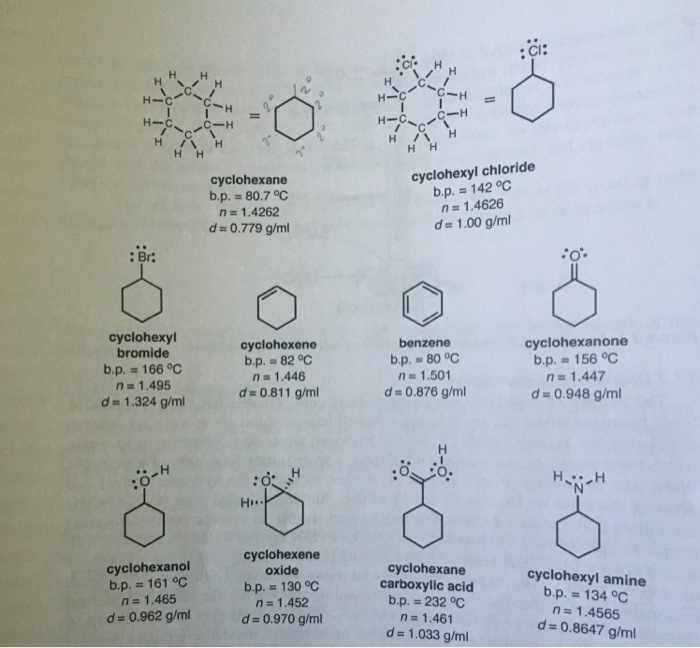
Solved Chair Conformation For Cyclohexane And Two Chair Chegg Com The chair conformation is estimated to be lower in energy than the twist conformation by approximately 23 kJ mol-1. Ive seen chair conformation defined in that way before. The molar mass of cyclohexane is 8416 g mol. Benzene has sp2 hybridized carbon atoms and cyclohexane has sp3 hybridised carbon atoms. Cyclic alkanes can also interconvert between their conformers by rotation of the single carbon to carbon bonds. In each of the conformations drawn in step 4 circle the axial substituents other than hydrogen.
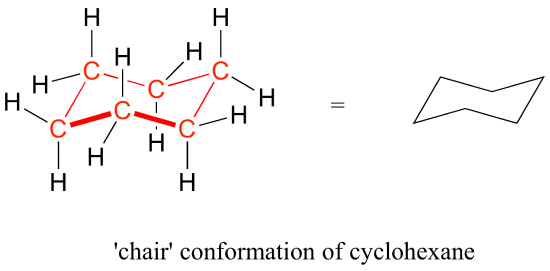 Source: chem.libretexts.org
Source: chem.libretexts.org
3 3 Conformations Of Cyclic Organic Molecules Chemistry Libretexts This group of compounds was called aromatic because of their pleasant odors. Cyclohexane has chair conformation. We can think of it as two chains mirror images one of the other containing atoms 1-2-3-4 and 1-6-5-4 with opposite dihedral angles. Cyclic alkanes can also interconvert between their conformers by rotation of the single carbon to carbon bonds. Identify the up tip OR down tip of your chair conformation and draw a straight line up up tip or down down tip parallel to the y-plane. The chair conformation of cyclohexane is not rigid.
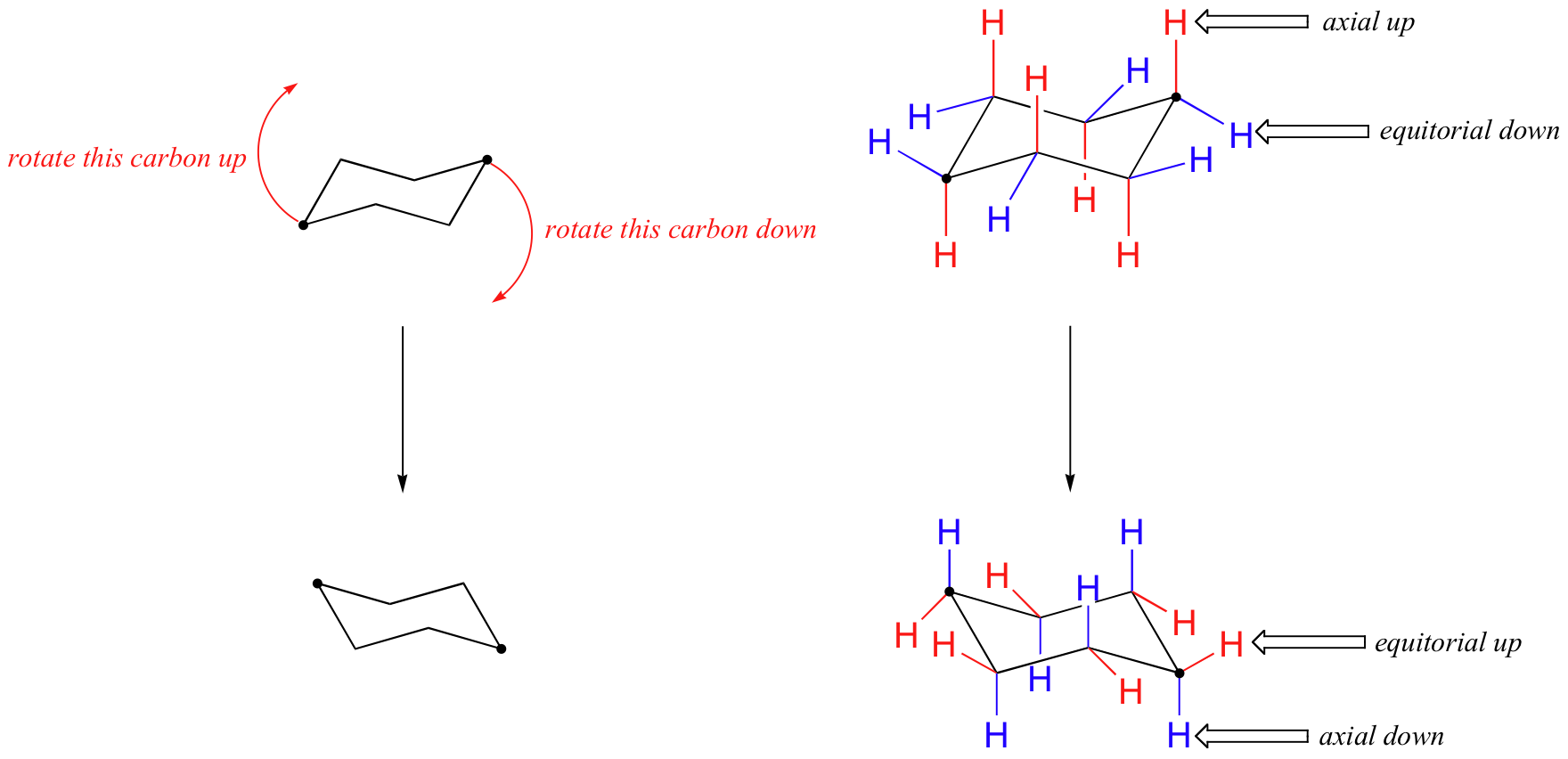 Source: chem.libretexts.org
Source: chem.libretexts.org
4 6 Axial And Equatiorial Bonds In Cyclohexane Chemistry Libretexts Get the drawing done in a third of the time so you can focus on the difficult parts of the reactions. The same term applies to similar conformations of six-member saturated ring analogous structures. Moreover the symmetry of chair conformation is D 3d while boat symmetry has the symmetry C 2v. Benzene is a planar structure. Boat and Twist Boat Conformations There are additional conformations of cyclohexane rings. The energy barriers between the chair boat and twist conformations of cyclohexane are low enough Fig6 to make separation of the.
Please support us by sharing this posts to your own social media accounts like Facebook, Instagram and so on or you can also save or be able to bookmark this blog page in this website. Thank you …










